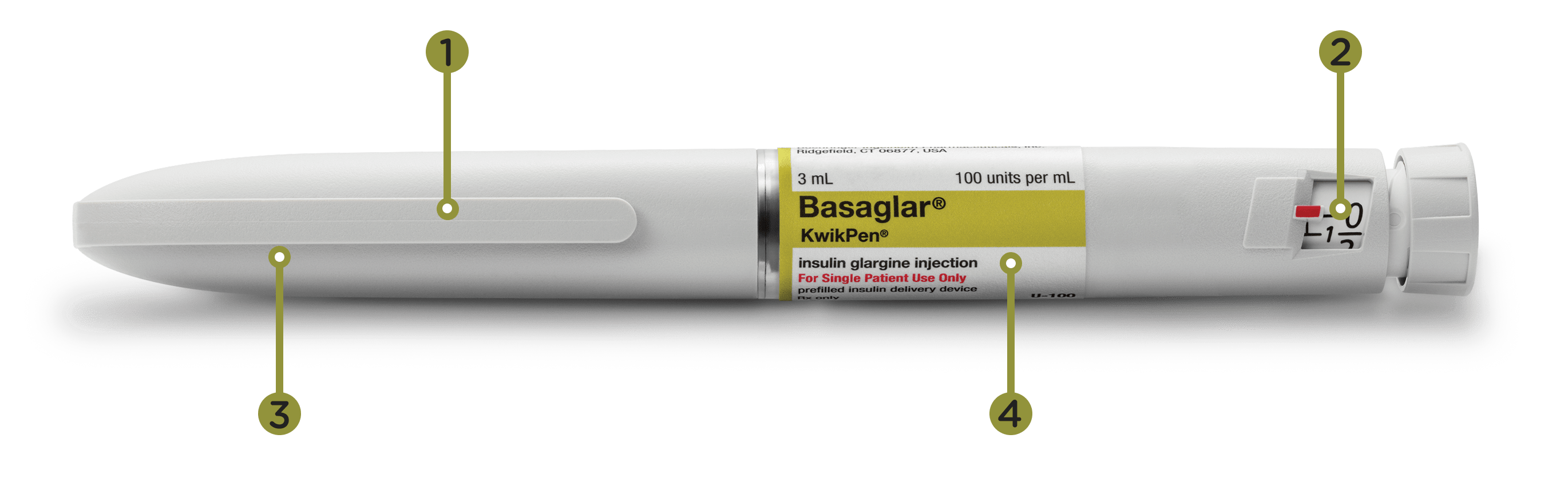
00:00-00:10
[Acoustic guitar music plays in background]
Caption: Basaglar® (insulin glargine) injection 100 Units/mL
Narrator: Basaglar is a long-acting insulin taken once a day, at the same time every day.
00:10-00:26
[A man and woman sit on a living room couch together; woman touches man’s arm; man smiles, nods, and gets up from the couch. In the kitchen, he opens refrigerator door]
Narrator: It's time to begin injecting Basaglar. Let's dive right in. You should always store unused Basaglar KwikPens in the refrigerator at 36 degrees Fahrenheit to 46 degrees Fahrenheit, or two degrees Celsius to eight degrees Celsius.
Caption: Unused pens need to be refrigerated
00:26-00:37
[Man stands in front of the open refrigerator, then closes it and walks away from refrigerator]
Narrator: Get your new Pen from the refrigerator each time you need one. The Pen does not need to be refrigerated after first use.
Caption: One Pen contains multiple doses of insulin
00:37-00:47
Narrator: One Pen contains multiple doses of insulin. Don't freeze Basaglar and don't use if it has been frozen. Unused pens may be used until the expiration date printed on the label if the Pen has been kept in the refrigerator.
Caption: Don’t freeze Basaglar
Caption: Unused Pens may be used until the expiration date printed on the label
00:47-00:59
[Image of stairs; man walks upstairs]
Narrator: You shouldn't reuse or share needles with another person. Don't share your Pen with anyone else, even if the needle is changed, because you may give them a serious infection or get a serious infection from them.
Caption: Don’t reuse or share Needles with another person
Caption: Don’t share your Pen with anyone else, even if the Needle is changed
Caption: You may give them a serious infection or get a serious infection from them
00:59-01:11
[The man walks down the hallway of his home; he washes his hands in his bathroom]
Narrator: This Pen isn't recommended for use by the blind or visually impaired without the help of someone trained to use the Pen. Remember to always wash your hands with soap and water before you inject.
Caption: This Pen isn’t recommended for use by the blind or visually impaired without the help of someone trained to use the Pen
01:11-01:22
[The man sits on his bed]
Narrator: Now that you have your Pen, make sure it's the right type of insulin and check the expiration date. This is especially important if you use more than one type of insulin.
Caption: Right type of insulin
Caption: Check expiration date
01:22-01:34
[The man is sitting on his bed; he picks up his Pen off the nightstand; Image of Pen, new needle in packaging, and alcohol swab in packaging]
Narrator: Do not use your Pen past the expiration date on the label. And remember, don't use your Pen if any parts look broken or damaged. Always keep an extra Pen in case yours is lost or damaged.
Caption: Don’t use if broken or damaged
Caption: Always keep extra Pen
01:34-01:48
[Image of man looking to his right; image of sterile needle; image of alcohol swab; image of Pen + sterile needle + alcohol swab]
Narrator: It's time to gather the rest of your supplies. Get a new needle and an alcohol swab. Remember to always use a new needle for each injection to help prevent infections and prevent blocked needles.
Caption: New Needle
Caption: Alcohol swab
01:48-02:04
Narrator: Now let's review these five sections and you'll be ready to inject in no time. First, prepare your Pen. Then, prime your Pen. Next, select your dose. Then, inject yourself. And to finish, clean up after your injection.
Caption: Prepare Your Pen
Caption: Prime Your Pen
Caption: Select Your Dose
Caption: Inject Yourself
Caption: Clean Up
02:04-02:19
[Image of green check mark with “Do” above and an image of a red X with “Don’t” above; the man is sitting on his bed and is pulling the Pen cap off his Pen]
Narrator: In this video, you'll see do’s in a green check mark and don'ts in a red cross out like this. Now it's time to prepare your Pen. Begin by pulling the Pen cap straight off.
Caption: Prepare Your Pen
Caption: Pull the Pen Cap straight off
02:19-02:31
[Image of a hand holding a Pen]
Narrator: If you can't remove the Pen cap, gently twist the cap back and forth and then pull it straight off. Don't remove the Pen label. This is a demonstration Pen. Please check your Pen for the label.
Caption: If you can’t remove the Pen Cap:
-
Gently twist back and forth
-
Pull it straight off
Caption: Don’t remove the Pen Label
Caption: This is a demonstration Pen, please check your Pen for the Label
02:31-02:46
[Image of a hand wiping the end of the Pen with an alcohol swab; image of Pen with clear, colorless liquid; image of Pen with cloudy, blue-tinted liquid]
Narrator: Now wipe the rubber seal with the alcohol swab. The liquid in the Pen should be clear and colorless. If it's not, begin again with a new Pen. Don't use if it's cloudy, colored, or has particles or clumps in it.
Caption: Check the liquid
Caption: Don’t use if it’s cloudy, colored, or has particles or clumps in it
02:46-02:59
[Image of hands pulling off the paper tab from the outer needle shield; image of man’s hands twisting the needle onto the Pen]
Narrator: Now select the new needle and pull off the paper tab from the outer needle shield. Push the capped needle straight onto the Pen and then twist the needle on until it's tight.
Caption: Select new Needle and remove tab
Caption: Push capped Needle straight onto Pen
Caption: Twist Needle until it’s tight
02:59-03:18
[Image of man looking at his Pen]
Narrator: Pull off both the outer and inner needle shields. Make sure you keep the outer needle shield but throw away the inner needle shield. Remember, you can always take a break and review these instructions. Take your time. You're all done preparing your Pen. Nice job. Let's keep going.
Caption: Pull off both Needle Shields
03:18-03:36
[Image of Pen, with arrows to show air moving out of the needle and cartridge]
Narrator: Now it's time to prime your Pen, which means we're moving the air from the needle and cartridge that may collect during normal use and ensures the Pen is working correctly. If you don't prime your Pen before each injection, you may get too much or too little insulin.
Caption: Prime Your Pen
03:36-03:50
[Image of hands holding Pen, turning the dose knob to two units; image of hands holding Pen with the needle point up, gently tapping the cartridge holder]
Narrator: Begin by turning the dose knob to select two units. Hold the Pen with the needle point up. Gently tap the cartridge holder so air bubbles collect at the top. Keep holding the Pen with the needle pointing straight up.
Caption: Select 2 units
Caption: Hold Pen upright and tap
03:50-04:02
[Hands push in the dose knob of the Pen; “0” is seen in the dose window; continuing to hold the Pen upright, hands hold the dose knob in; image of upright Pen, with a drop of insulin at the tip of the needle]
Narrator: Now push the dose knob in until it stops and “0” is seen in the dose window. Hold the dose knob in and slowly count to five. You should see insulin at the tip of the needle.
Caption: Slowly count to 5
04:02-04:16
[Image of small air bubbles in cartridge]
Narrator: If you don't see insulin, repeat the priming steps, but not more than four times. If you still don't see insulin, change the needle and repeat the priming steps. Small air bubbles are normal and won't affect your dose.
Caption: Repeat Priming steps until you see insulin at tip
Caption: Air bubbles
04:16-04:28
[Image of two hands holding Pen: the left hand holds the Pen while the right hand turns the dose knob; image of hand holding Pen horizontally in one hand, with the selected dose appearing in the dose knob window]
Narrator: Another one done. Time to select your dose. Begin by turning the dose knob to select the number of units you need to inject. The dose indicator should line up with your dose.
Caption: Select Your Dose
Caption: Turn Dose Knob to units you need to inject
04:28-04:46
[Hands turn the dose knob; image of hands turning the dose knob in both directions]
Narrator: The Pen dials one unit at a time, and the dose knob clicks as you turn it. Don't dial your dose by counting the clicks because you may dial the wrong dose. The dose can be corrected by turning the dose knob in either direction until the correct dose lines up with the dose indicator.
Caption: Don’t dial your dose by counting the clicks because you may dial the wrong dose
04:46-5:02
[Image of dose knob pulled out all the way, with hands turning the knob in either direction to show the numbers on the side of the Pen]
Narrator: The even numbers are printed on the dial, and the odd numbers after the number “1” are shown as full lines. Always check the number in the dose window to make sure you have dialed the correct dose. You can only dial the number of units left in the Pen.
05:02-05:13
[Image of upright Pen]
Narrator: The Pen can hold 300 units of insulin and can deliver up to 80 units in one injection. The cartridge contains an additional small amount of insulin that can't be delivered.
Caption: Total 300 units
Caption: 80 units at a time
05:13-5:26
[Image of two pens (one is open, and one is closed), with a + sign between them]
Narrator: If your dose is more than the number of units left in the Pen, you may either inject the units left in your Pen and then use a new Pen for the rest of your dose or use a new Pen for the full dose.
Caption: Units left
05:26-05:40
Narrator: Here are a few things to remember if the dose knob is hard to push. Pushing the dose knob slowly will make it easier to inject. Your needle may be blocked. Put on a new needle and prime the Pen.
Caption: If Dose Knob is hard to push:
-
Push Dose Knob Slowly
-
Put on new Needle and prime Pen
-
Throw Pen away and replace it
05:40-05:51
[The man is sitting on his bed, holding his Pen, which he is ready to inject]
Narrator: You may have dust, food, or liquid inside the Pen. Throw the Pen away and get a new one. You're already halfway through. Now it's time to inject.
05:51-6:01
Narrator: Remember, you can always go back if you need more time to review the steps. No pressure. Here are a few things to keep in mind while injecting.
Caption: Inject Yourself
6:01-06:25
Narrator: Inject your insulin as your healthcare provider has shown you. Change (rotate) your injection sites within the area you choose for each dose to reduce your risk of getting lipodystrophy (pits and skin or thickened skin) and localized cutaneous amyloidosis (skin with lumps) at the injection sites. Do not inject where the skin has pits, is thickened, or has lumps.
Caption: Reminders:
-
Inject your insulin as your healthcare provider has shown you
-
Change your injection site for each injection
-
Don’t try to change your dose while injecting
06:25-6:38
[The man is sitting on his bed, preparing to inject the Pen]
Narrator: Do not inject where the skin is tender, bruised, scaly or hard, or into scars or damaged skin. Don't try to change your dose while injecting. First, choose your injection site.
06:38-06:52
[Image of man’s body, from his shoulders to just above his knees; on the man’s body, areas where Basaglar can be injected are highlighted in blue; the man is sitting on his bed; he wipes an area of his stomach with an alcohol swab]
Narrator: Basaglar is injected under the skin of your stomach area, upper legs, upper arms, or buttocks. Wipe the skin with an alcohol swab and let the injection site dry before you inject.
Caption: Choose your injection site
-
Stomach
-
Upper Legs
-
Upper Arms
-
Buttocks
Caption: Wipe with an alcohol swab
06:52-07:06
[The man inserts the needle into the skin of his stomach; Image of the man inserting the needle into his stomach while a timer counts from 1 to 5]
Narrator: Insert the needle into your skin. Put your thumb on the dose knob and push the dose knob in until it stops. Hold the dose knob in and slowly count to five.
Caption: Insert Needle
Caption: Push Dose Knob in
Caption: Hold Dose Knob in and count to 5
07:06-07:17
[The man pulls the needle out of his skin]
Narrator: Don't try to inject your insulin by turning the dose knob. You will not receive your insulin by turning the dose knob. Now pull the needle straight out of your skin.
Caption: Don’t try to inject your insulin by turning the Dose Knob
Caption: You will not receive your insulin by turning the Dose Knob
Caption: Pull Needle out
07:17-07:34
[Image of Pen with a drop of insulin at the tip of the needle; image of Pen showing “0” in the dose window]
Narrator: A drop of insulin at the needle tip is normal. It won't affect your dose. If you see “0” in the dose window, you've received the full amount you dialed. If you don't see “0” in the dose window, don't redial. Insert the needle into your skin and finish your injection.
Caption: Check Dose Window
07:34-07:46
[Image of two pens; image of plunger inside Pen moving up, with a blue arrow on the left side of the Pen]
Narrator: If you still don't think you received the full amount you dialed for your injection, don't start over or repeat that injection. Keep track of your blood sugar and call your healthcare provider for further instructions.
Caption: Don’t start over or repeat that injection
07:46-07:58
Narrator: If you normally need two injections for your full dose, be sure to take your second injection. The plunger only moves a little with each injection, so you may not notice that it moves.
07:58-08:11
[Image of alcohol swab on skin]
Narrator: If you see blood after you take the needle out of your skin, just press the spot lightly with a piece of gauze or an alcohol swab. Don't rub the area. And that's how you inject Basaglar.
Caption: Press lightly with alcohol swab
08:11-08:25
[Image of hand dropping a Pen into a sharps container; image of hand putting the Pen into an outer needle shield, unscrewing the capped needle, and throwing it away in a sharps container]
Narrator: And last but not least, it's time to clean up after your injection. Carefully replace the outer needle shield, unscrew the capped needle, and throw it away in a sharps container.
Caption: Clean Up
Caption: Replace Outer Needle Shield
Caption: Unscrew capped Needle
Caption: Throw it away in a sharps container
08:25-08:39
[Image of Pen with needle still attached and a red “X” over the needle; the man holds the Pen and replaces the Pen cap]
Narrator: Don't store the Pen with the needle attached. Remove the needle to prevent leaking, blocking of the needle, and air from entering the Pen. Replace the Pen cap by lining up the cap clip with the dose indicator and pushing straight on.
Caption: Replace Pen Cap
08:39-08:50
[The man puts his used needle in a sharps container; he then walks into his laundry room and picks up a laundry detergent container]
Narrator: Put your used needles in an FDA-cleared sharps disposal container right away after use. Don't throw away loose needles in your household trash.
08:50-09:11
Narrator: If you don't have an FDA-cleared sharps disposal container, you may use a household container that's made of heavy-duty plastic, can be closed with a tight fitting, puncture-resistant lid without sharps being able to come out, upright and stable during use, leak resistant, and properly labeled to warn of hazardous waste inside the container.
Caption:
-
Heavy-duty plastic
-
Puncture-resistant lid
-
Upright and stable
-
Leak resistant
-
Properly labeled
09:11-09:24
[The man walks downstairs; he sits down at his home office desk and opens his computer]
Narrator: When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used needles and syringes.
09:24-09:38
Narrator: For more information about safe sharps disposal and for specific information about sharps disposal in the state that you live in, go to the FDA's website at www.fda.gov/safesharpsdisposal.
09:38-09:49
[Image of trash bin crossed out; image of recycle bin crossed out]
Narrator: And remember, don't dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Don't recycle your used sharps disposal container either.
Caption: Don’t dispose in household trash
Caption: Don’t recycle
09:49-10:04
[Man places his Pen in a small box and places a lid on the box, then places the box on a high shelf]
Narrator: Make sure to store the Pen you're currently using in a safe place at room temperature (up to 86 degrees Fahrenheit or 30 degrees Celsius), away from heat and light. And always keep the Pen you're using in a spot where kids can't reach it.
10:04-10:16
[Image of calendar that flips from 1 day to 28 days; a hand throws a used Pen into a household trash can]
Narrator: The Pen you're currently using should be thrown away after 28 days even if it still has insulin left in it. Your used Pen can be thrown away in your household trash after you've removed the needle.
Caption: The Pen you’re currently using should be thrown away after 28 days even if it still has insulin in it
10:16-10:34
[The man walks down the hallway in his home; he glances at a photo of two children as he walks by it; he returns to the living room couch, where he sits down next to his wife; he puts his arm around her, and she puts her hand on his chest]
Narrator: Here are a few things to remember. Always use a new needle for each injection. Don't share your Pen or needles with other people. You may give them a serious infection or get a serious infection from them. Keep your Pen and needles out of sight and reach of children.
Caption: Always use a new Needle for each injection
Caption: Don’t share your Pen or Needles with other people. You may give them a serious infection or get a serious infection from them
Caption: Keep your Pen and Needles out of sight and reach of children
10:34-10:51
Narrator: Nice job. You're all done. Now go ahead and clean up your supplies so they're all set for next time. That's all, folks. Watch this video and check out the full instructions for use that came with your Basaglar KwikPen anytime you're unsure of a step, and you'll be injecting in no time. We're here to help.
Caption: All done!
Caption: This video must be used with the complete Instructions for Use included with the Pen
10:51-11:11
Narrator: If you have any questions about your Basaglar KwikPen, contact Lilly at 1-800LillyRx. That's 1-800-545-5979 or call your health care provider.
Caption: If you have questions, contact Lilly at 1-800LillyRx (1-800-545-5979) or call your healthcare provider
Caption: Basaglar®
(insulin glargine) injection
100 Units/mL
Caption: PP-BV-US-1940 11/2022 ©Lilly USA, LLC 2022. All rights reserved.
BASAGLAR is a registered trademark owned or licensed by Eli Lilly and Company, its subsidiaries or affiliates.
KwikPen is a registered trademark of Eli Lilly and Company.
BD Ultra-Fine is a trademark of Becton, Dickinson and Company.